|
Most common psychiatric comorbidities found to be associated with autism include anxiety and intellectual disability. The current emphasis on autism spectrum disorders has generated new opportunities in the field of neuroscience, and further advancements in the identification of different biomarkers, risk genes, and genetic pathways can help in the early diagnosis and development of new clinical and pharmacological treatments for ASD.Īutism spectrum disorder (ASD) is a complex neurological disorder that affects an individual’s development by impairing social interaction and communication and causes stereotypical behaviors that disrupt the anatomy and functional connectivity in the brain. This review highlights the role of ASD risk genes in gene transcription and translation regulation processes, as well as neuronal activity modulation, synaptic plasticity, disrupted key biological signaling pathways, and the novel candidate genes that play a significant role in the pathophysiology of ASD. Identification of ASD risk genes helps to unravel various genetic variants and signaling pathways which are involved in ASD. The considerable significance of the genetics associated with autism has led to the identification of many risk genes for ASD used for the probing of ASD specificity and shared cognitive features over the past few decades. This knowledge is important for understanding complex brain activities in general, but also to improve the treatment of neurological disorders such as DS.Autism spectrum disorder (ASD) is a genetic heterogeneous neurodevelopmental disorder that is characterized by impairments in social interaction and speech development and is accompanied by stereotypical behaviors such as body rocking, hand flapping, spinning objects, sniffing and restricted behaviors. My experience on measuring subcellular axonal functions as well as neuronal network oscillations provides the unique possibility to advance our understanding of the causal relationship between axonal currents, AP signalling and GABAergic inhibition in neuronal networks. So far, other studies were unable to directly resolve the ion channel activity in PV-IN axons of DS mice because of the difficulties in accessing the fine axonal structures. The project aims to identify how axonal functions are impaired in DS mice and to link the axonal deficits to dysfunctions in neuronal network oscillations. These dysfunctions are believed to be caused by changes in action potential (AP) signalling in PV-IN axons. In close agreement, transgenic mice with reduced SCN1A gene expression (DS mice) reproduce key features of DS symptoms and show dysfunctions in parvalbumin-expressing GABAergic interneurons (PV-INs), as well as deficits in cortical network oscillations. 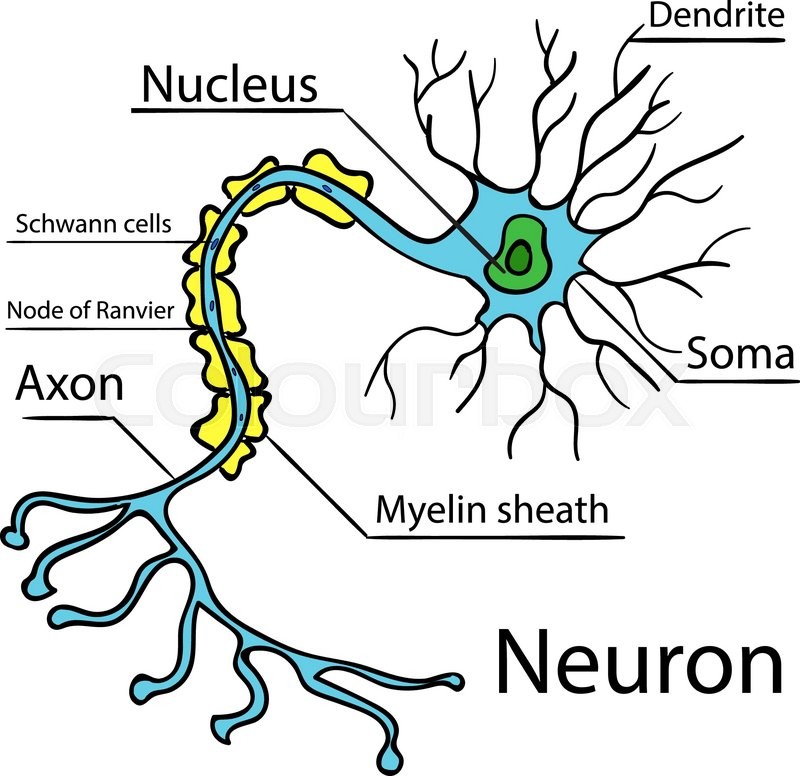
Impairments of GABAergic inhibition are known to lead to epilepsy and other cognitive disorders. These inhibitory neurons are responsible for balancing cortical brain activity by the release of the neurotransmitter GABA from their axonal terminals. 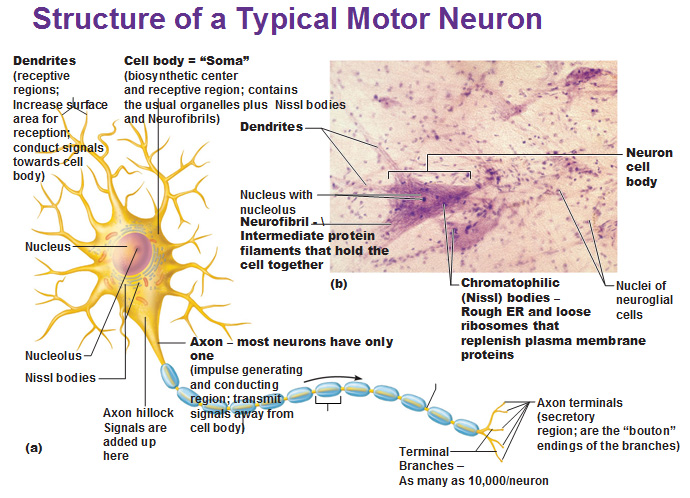
Most DS patients show a mutation in the SCN1A gene, which encodes for a sodium channel subunit (NaV1.1) highly expressed in axons of cortical GABAergic interneurons. Dravet Syndrome (DS) is a severe form of childhood epilepsy with an early onset (under one year) affecting cognitive development and often causing sudden death.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
